Influence of Mass Variation of Green-Synthesized CoFe2O4/Cdots Catalyst on the Photocatalytic Degradation of Methylene Blue Through Initial Adsorption Mechanism
DOI:
https://doi.org/10.22437/j-bigme.v6i1.49932Keywords:
Photocatalyst, Adsorption, CoFe2O4/Cdots, Methylene Blue DegradationAbstract
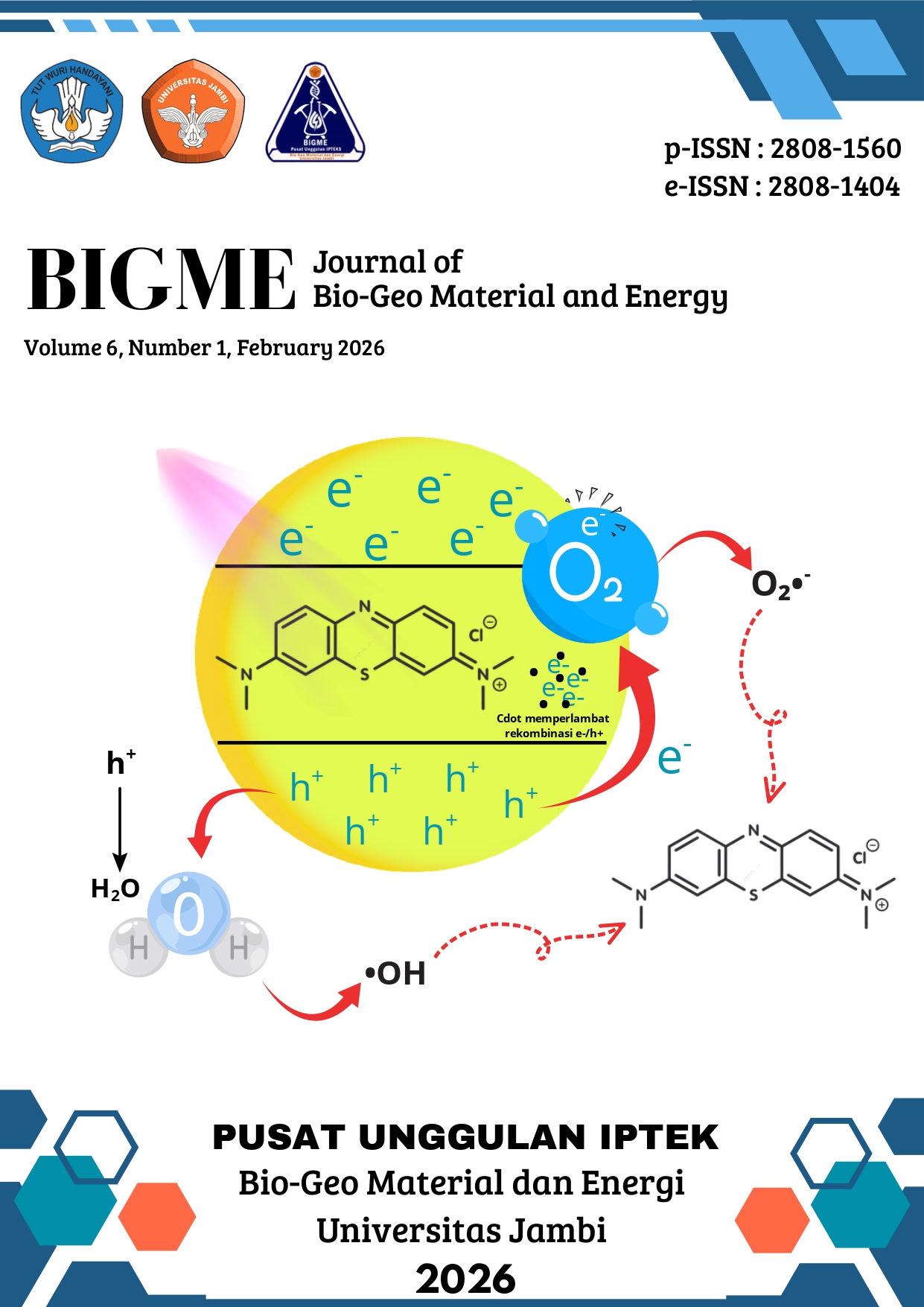
This study aimed to synthesize and evaluate the photocatalytic performance of CoFe₂O₄/Cdots nanocomposites prepared via green synthesis method using Moringa oleifera leaf extract and watermelon rind waste as eco-friendly precursors. The synthesis involved a co-precipitation process for CoFe₂O₄ formation and a hydrothermal route to produced Cdots, followed by composite fabrication. The materials were characterized using XRD, UV-Vis Spectrophotometer, and VSM analyses to determine their crystalline structure, optical, and magnetic properties. The results showed that the incorporation of Cdots increased the optical band gap (3.15 eV) compared to pure CoFe₂O₄ (2.64 eV) and improved soft magnetic behavior. Photocatalytic tests under UV irradiation using Methylene Blue as a model pollutant revealed a degradation efficiency of 91.9% for CoFe₂O₄/Cdots at an optimum catalyst mass of 0.07 gr, significantly higher than 60,8% achieved by pure CoFe₂O₄. The enhanced performance is attributed to the synergistic interaction between CoFe₂O₄ and Cdots, which promotes charge separation, initial adsorption, and reactive radical generation. These findings indicate that the CoFe₂O₄/Cdots nanocomposite is a promising eco-friendly photocatalyst for wastewater treatment applications.
Downloads
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Aldwin Zalukhu, Frastica Deswardani, Edi Suharyadi, Ranti Widia Utami, Nurul Imani Istiqomah, Larrisa Jestha Mahardhika, Siti Fatimah Azzahro, Dapfa Farrizo Altop, Nada Oktaviani

This work is licensed under a Creative Commons Attribution 4.0 International License.