Anticaries Potential of Temu Kunci-Serai Ethyl Acetate Extract Combination: In Vitro and Molecular Studies Approach
DOI:
https://doi.org/10.22437/chp.v7i1.26098Keywords:
temu kunci, serai, molecular docking, Streptococcus mutans, 3AICAbstract
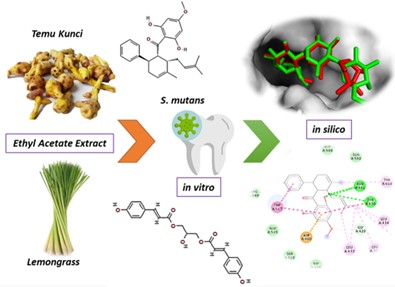
Dental caries is one of the problems in dental disorders suffered by many people. There have been many ways of handling such as using temu kunci and lemongrass plants. This study aims to determine the antibacterial activity of caries in vitro and predict the mechanism of action of the bacteria that cause caries Streptococcus mutans.  Temu kunci and lemongrass were extracted using ethyl acetate solvent. antibacterial tests against Streptococcus mutans were carried out using diffusion tests with a combination treatment of temu kunci: serai extracts 5% b/v: 5% b/v, 5% b/v: 10% b/v, and 10% b/v: 5% b/v. The positive control used amoxicillin and DMSO as a negative control. Potency as an anticaries drug of drived compound from Temu Kunci and Serai were evaluated by molecular docking using glucosyltransferase (3AIC). The results showed that the combination of temu kunci and serai with concentration 10% b/v: 5% b/v has potential as anticaries against Streptococcus mutans. Molecular studies depicted that Panduratin A, Isopanduratin, and 1,3-O-di-p-coumaroylglycerol have great activity toward 3AIC, respectively. Especially for Panduratin A and Isopanduratin, those compound depicted great and similar binding affinity (-8.4 kcal mol-1) that lower than Acarbose as native ligand  (-8.3 kcal mol-1). Furthermore, those compound binding similarity ilustrated activity mechanism similarly with native ligand toward receptor. Additionally, the profiling drug-target interaction suggested Temu Kunci’s derived compounds have great potential as anticaries treatment.  Â
Downloads
References
1. El Sherbiny, G. M. (2014). Control of growth Streptococcus mutans isolated from saliva and dental caries. Ournal of Current Microbiology and Applied Sciences, 3(10), 1–10.
2. Lemos, J. A., Palmer, S. R., Zeng, L., Wen, Z. T., Kajfasz, J. K., Freires, I. A., Abranches, J., & Brady, L. J. (2019). The Biology of Streptococcus mutans. Microbiology Spectrum, 7(1). https://doi.org/10.1128/microbiolspec.GPP3-0051-2018
3. World Health Organization. (2019, June 13). Ending childhood dental caries: WHO implementation manual. Geneva: World Health Organization. https://apps.who.int/iris/handle/10665/330643
4. Kementerian Kesehatan RI. (2019). Riset Kesehatan Dasar 2018.
5. Pitts, N. B., Zero, D. T., Marsh, P. D., Ekstrand, K., Weintraub, J. A., Ramos-Gomez, F., Tagami, J., Twetman, S., Tsakos, G., & Ismail, A. (2017). Dental caries. Nature Reviews Disease Primers, 3(1), 17030. https://doi.org/10.1038/nrdp.2017.30
6. Yon, M. J. Y., Gao, S. S., Chen, K. J., Duangthip, D., Lo, E. C. M., & Chu, C. H. (2019). Medical Model in Caries Management. Dentistry Journal, 7(2), 37. https://doi.org/10.3390/dj7020037
7. Chahyadi, A., Hartati, R., Wirasutisna, K. R., & Elfahmi. (2014). Boesenbergia Pandurata Roxb., An Indonesian Medicinal Plant: Phytochemistry, Biological Activity, Plant Biotechnology. Procedia Chemistry, 13, 13–37. https://doi.org/10.1016/j.proche.2014.12.003
8. Do, D. N., Nguyen, D. P., Phung, V.-D., Le, X.-T., Le, T. M., Do, V. M., Minh, B. Q., & Luu, X. C. (2021). Fractionating of Lemongrass (Cymbopogon citratus) Essential Oil by Vacuum Fractional Distillation. Processes, 9(4), 593. https://doi.org/10.3390/pr9040593
9. Oladeji, O. S., Adelowo, F. E., Ayodele, D. T., & Odelade, K. A. (2019). Phytochemistry and pharmacological activities of Cymbopogon citratus: A review. Scientific African, 6, e00137. https://doi.org/10.1016/j.sciaf.2019.e00137
10. Hwang, J.-K., Shim, J.-S., & Chung, J.-Y. (2004). Anticariogenic activity of some tropical medicinal plants against Streptococcus mutans. Fitoterapia, 75(6), 596–598. https://doi.org/10.1016/j.fitote.2004.05.006
11. Taweechaisupapong, S., Singhara, S., Lertsatitthanakorn, P., & Khunkitti, W. (2010). Antimicrobial effects of Boesenbergia pandurata and Piper sarmentosum leaf extracts on planktonic cells and biofilm of oral pathogens. Pakistan Journal of Pharmaceutical Sciences, 23(2), 224–231.
12. Ortega Cuadros, M., Tofiño Rivera, A. P., Merini, L. J., & Martinez Pabon, M. C. (2018). Antimicrobial activity of Cymbopogon citratus (Poaceae) essential oil on Streptococcus mutans biofilm and cytotoxic effect on keratinocytes and fibroblasts. Revista de BiologÃa Tropical, 66(4), 1519. https://doi.org/10.15517/rbt.v66i4.33140
13. Hati, A. K., Dyahariesti, N., & Yuswantina, R. (2019). Penetapan Kadar Flavonoid Dan Uji Aktivitas Antibakteri Ekstrak Sereh (Cymbopogon nardus) Dan Temu Kunci (Boesnbergia pandurata Roxb) Terhadap Bakteri Streptococcus Mutans. Indonesian Journal of Pharmacy and Natural Product, 2(2), 71–78. https://doi.org/10.35473/ijpnp.v2i2.264
14. Kawengian, S. A. F., Wuisan, J., & Leman, M. A. (2017). Uji daya hambat ekstrak daun serai (Cymbopogon citratus L) terhadap pertumbuhan Streptococcus mutans. E-GIGI, 5(1), 7–11. https://doi.org/10.35790/eg.5.1.2017.14736
15. Elfahmi, Aziz, S., & Sulaiman, I. (2017). Isolation of flavonoid compound from ethyl acetate extract of fingeroot (Boesenbergia Pandurata (Roxb.) Schlechter) rhizome. Research Journal of Pharmaceutical, Biological and Chemical Sciences, 8(4), 1192–1198.
16. Priyadi, M., Chusna, N., Isnawati, I., & Indriani, O. (2021). Profil Fitokimia Ekstrak Etil Asetat Temu Kunci (Boesenbergia rotunda L.) dan Serai (Cymbopogon citratus). Jurnal Pharmascience, 8(1), 45. https://doi.org/10.20527/jps.v8i1.9725
17. Adhikari, D., Gong, D.-S., Oh, S. H., Sung, E. H., Lee, S. O., Kim, D.-W., Oak, M.-H., & Kim, H. J. (2020). Vasorelaxant Effect of Boesenbergia rotunda and Its Active Ingredients on an Isolated Coronary Artery. Plants, 9(12), 1688. https://doi.org/10.3390/plants9121688
18. Atun, S., Sri, H., & Rakhmawati, A. (2018). Potential Bioactive Compounds Isolated from Boesenbergia rotunda as Antioxidant and Antimicrobial Agents. Pharmacognosy Journal, 10(3), 513–518. https://doi.org/10.5530/pj.2018.3.84
19. Song, M. S., Shim, J. S., Gwon, S. H., Lee, C. W., Lee, C. W., Kim, H. S., & Hwang, J. K. (2008). Antibacterial activity of panduratin A and isopanduratin A isolated from Kaempferia pandurata Roxb. against acne-causing microorganisms. Food Science and Biotechnology, 17(6), 1357–1360.
20. Rukayadi, Y., Lee, K.-H., & Hwang, J.-K. (2009). Activity of panduratin A isolated from Kaempferia pandurata Roxb. against multi-species oral biofilms in vitro. Journal of Oral Science, 51(1), 87–95. https://doi.org/10.2334/josnusd.51.87
21. Hwang, J.-K., Chung, J.-Y., Baek, N.-I., & Park, J.-H. (2004). Isopanduratin A from Kaempferia pandurata as an active antibacterial agent against cariogenic Streptococcus mutans. International Journal of Antimicrobial Agents, 23(4), 377–381. https://doi.org/10.1016/j.ijantimicag.2003.08.011
22. Hacke, A. C. M., Marques, J. A., Vellosa, J. C. R., Boligon, A. A., da Silva, F. D., de Souza, D., Bonini, J. S., Rocha, J. B. T., & Pereira, R. P. (2018). Ethyl acetate fraction of Cymbopogon citratus as a potential source of antioxidant compounds. New Journal of Chemistry, 42(5), 3642–3652. https://doi.org/10.1039/C7NJ04352J
23. Wang, H., Zhang, R., Zhang, K., Chen, X., & Zhang, Y. (2022). Antioxidant, Hypoglycemic and Molecular Docking Studies of Methanolic Extract, Fractions and Isolated Compounds from Aerial Parts of Cymbopogon citratus (DC.) Stapf. Molecules, 27(9), 2858. https://doi.org/10.3390/molecules27092858
24. Nijampatnam, B., Zhang, H., Cai, X., Michalek, S. M., Wu, H., & Velu, S. E. (2018). Inhibition of Streptococcus mutans Biofilms by the Natural Stilbene Piceatannol Through the Inhibition of Glucosyltransferases. ACS Omega, 3(7), 8378–8385. https://doi.org/10.1021/acsomega.8b00367
25. Atta, L., Khalil, R., Khan, K. M., Zehra, M., Saleem, F., Nur-e-Alam, M., & Ul-Haq, Z. (2022). Virtual Screening, Synthesis and Biological Evaluation of Streptococcus mutans Mediated Biofilm Inhibitors. Molecules, 27(4), 1455. https://doi.org/10.3390/molecules27041455
26. Jauhar, M. M., Syaifie, P. H., Arda, A. G., Ramadhan, D., Nugroho, D. W., Ningsih Kaswati, N. M., Noviyanto, A., Rochman, N. T., & Mardliyati, E. (2023). Evaluation of propolis activity as sucrose-dependent and sucrose-independent Streptococcus mutans inhibitors to treat dental caries using an in silico approach. Journal of Applied Pharmaceutical Science, 13(3), 071–080. https://doi.org/10.7324/JAPS.2023.45365
27. Performance Standards for Antimicrobial Susceptibility Testing, 28th ed, Pub. L. No. M100, Wayne, PA: Clinical and Laboratory Standards Institute 1 (2018).
28. sayed, H., issa, mohammady, mahmoud, magda, abdelkader, emad, & ismail, hamadi. (2022). Phytochemical Screening and Antioxidant Activity of some Egyptian Medicinal Plants. Journal of Modern Research, 4(1), 14–20. https://doi.org/10.21608/jmr.2021.83376.1075
29. Tadtong, S., Watthanachaiyingcharoen, R., & Kamkaen, N. (2014). Antimicrobial Constituents and Synergism Effect of the Essential Oils from Cymbopogon citratus and Alpinia galanga. Natural Product Communications, 9(2), 1934578X1400900. https://doi.org/10.1177/1934578X1400900237
30. Prince, A., Roy, S., & McDonald, D. (2022). Exploration of the Antimicrobial Synergy between Selected Natural Substances on Streptococcus mutans to Identify Candidates for the Control of Dental Caries. Microbiology Spectrum, 10(3), e02357-21. https://doi.org/10.1128/spectrum.02357-21
31. Zhang, G., Tan, Y., Yu, T., Wang, S., Liu, L., & Li, C. (2021). Synergistic antibacterial effects of reuterin and catechin against Streptococcus mutans. LWT, 139, 110527. https://doi.org/10.1016/j.lwt.2020.110527
32. Ali, H. I., Ashida, N., & Nagamatsu, T. (2008). Antitumor studies. Part 4: Design, synthesis, antitumor activity, and molecular docking study of novel 2-substituted 2-deoxoflavin-5-oxides, 2-deoxoalloxazine-5-oxides, and their 5-deaza analogs. Bioorganic & Medicinal Chemistry, 16(2), 922–940. https://doi.org/10.1016/j.bmc.2007.10.014
33. Syarafina, Z. Y. I., Safithri, M., Bintang, M., & Kurniasih, R. (2022). In Silico Screening of Cinnamon (Cinnamomum burmannii) Bioactive Compounds as Acetylcholinesterase Inhibitors. Jurnal Kimia Sains Dan Aplikasi, 25(3), 97–107. https://doi.org/10.14710/jksa.25.3.97-107
34. Prahasti, A. E., Yuanita, T., & Rahayu, R. P. (2022). Computer Aided Drug Discovery Utilization in Conservative Dentistry. Journal of International Dental and Medical Research, 15(2), 899–903.
35. Ariefin, M., & Saputra, R. R. (2023). Study Natural Compound of Eleutherine americana as a SaR-CoV-2 Therapeutic Agent : In Silico Approach. ALCHEMY:Journal of Chemistry, 11(1), 41–49. https://doi.org/10.18860/al.v11i1.18018
36. Arora, R., de Beauchene, I. C., Polanski, J., Laine, E., & Tchertanov, L. (2013). Raltegravir flexibility and its impact on recognition by the HIV-1 IN targets. Journal of Molecular Recognition, 26(9), 383–401. https://doi.org/10.1002/jmr.2277
37. Bare, Y., Sari, D. R. T., Rachmad, Y. T., Krisnamurti, G. C., & Elizabeth, A. (2019). In Silico Insight the Prediction of Chlorogenic Acid in Coffee through Cyclooxygenase-2 (COX2) Interaction. Biogenesis: Jurnal Ilmiah Biologi, 7(2), 100–105. https://doi.org/10.24252/bio.v7i2.9847
38. Kharisma, V. D., Kharisma, S. D., Ansori, A. N. M., Kurniawan, H. P., Witaningrum, A. M., Fadholly, A., & Tacharina, M. R. (2021). Antiretroviral effect simulation from black tea (Camellia sinensis) via dual inhibitors mechanism in HIV-1 and its social perspective in Indonesia. Research Journal of Pharmacy and Technology, 14(1), 455–460. https://doi.org/10.5958/0974-360X.2021.00083.4
Published
Versions
- 2026-02-24 (2)
- 2023-07-30 (1)