The Role of Macrocyclic Compounds as Supramolecular Drug Delivery Systems: A-Review
DOI:
https://doi.org/10.22437/chp.v9i1.41724Keywords:
Drug Delivery, macrocyclic, supramoleculesAbstract
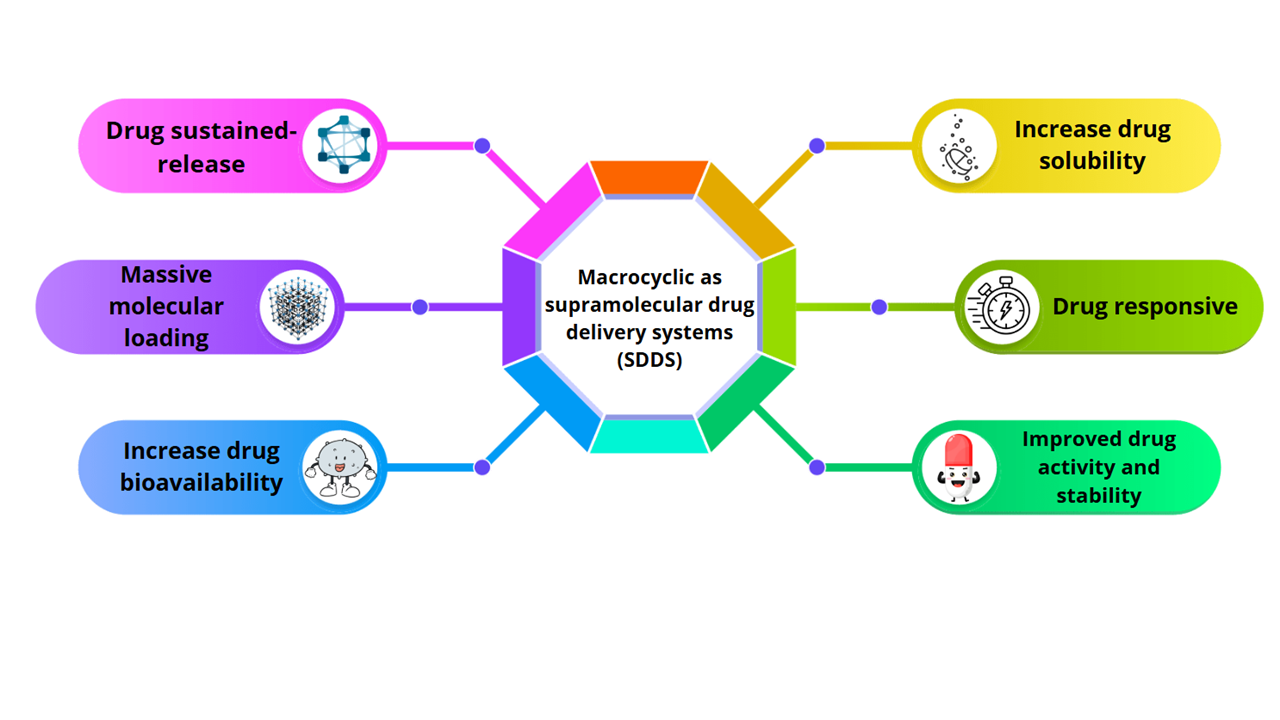
In recent decades, significant advances in the development of drug delivery systems have unlocked new possibilities for enhancing therapeutic efficacy. Among the diverse range of innovative materials, macrocyclic-based supramolecular systems have emerged as promising platforms due to their unique physicochemical properties. Macrocyclic compounds such as cyclodextrins and cucurbiturils exhibit a remarkable ability to form stable inclusion complexes with various drug molecules, thereby improving their solubility, chemical stability, bioavailability, and pharmacokinetic profiles. This review highlights the design principles, synthetic strategies, and mechanisms of action underlying macrocyclic drug carriers, with particular emphasis on their responsiveness to environmental stimuli such as pH, temperature, and biomolecular triggers. Recent findings demonstrate that macrocyclic systems can significantly enhance drug loading efficiency, targeted delivery, and cellular uptake, while minimizing systemic toxicity. These advances underscore the potential of macrocyclic supramolecules as foundational elements for the development of next-generation drug delivery systems that are more precise, effective, and adaptable to personalized therapeutic needs.
Downloads
References
[1]. Deng, J. H., J. Luo, Y.-L. Mao, S. Lai, Y.-N. Gong, D.-C. Zhong, T.-B. Lu, π-π stacking interactions: Non-negligible forces for stabilizing porous supramolecular frameworks. Science Advances. 2020;6(2), eaax9976. https://doi.org/10.1126/sciadv.aax9976
[2]. Tong, F., Zhou, Y., Xu, Y., Chen, Y., Yudintceva, N., Shevtsov, M & Gao, H. Supramolecular nanomedicines based on host-guest interactions of cyclodextrins. Exploration (Beijing). 2023;3(4)20210111. https://doi.org/10.1002/EXP.20210111
[3]. Zhou, J., Yu, G & Huang, F. Supramolecular chemotherapy based on host–guest molecular recognition: a novel strategy in the battle against cancer with a bright future. Chemical Society Reviews. 2017;46. https://doi.org/10.1039/C6CS00898D
[4]. Wu, J. R., Wu, G & Yang. Y. W. Pillararene-Inspired Macrocycles: From extended Pillar[ n]arenes to Geminiarenes. Accounts of Chemical Research, 2022;55(21). https://doi.org/10.1021/acs.accounts.2c00555.
[5]. Wu, D., Wang, J., Du, X., Cao, Y., Ping, K & Liu, D. Cucurbit[8]uril-based supramolecular theranostics. Joirnal of Nanobiotechnology, 2024;9(22),235). https://doi.org/10.1186/s12951-024-02349-z
[6]. Yang, Y., Li, P., Feng, H., Zeng, R., Li, S & Zhang, Q. Macrocycle-Based Supramolecular Drug Delivery Systems: A Concise Review. Molecules, 2024;29, 3828. https://doi.org/10.3390/molecules29163828
[7]. Webber, M, J., & Langer, R. Drug delivery by supramolecular design. Chemical Society Review, 2017;46, 6600. https://doi.org/10.1039/C7CS00391A
[8]. Geng, W. C., Jiang, Z. T., Chen, S. L & Guo, D. S. Supramolecular interaction in the action of drug delivery systems. Chemical Sciences, 2024;15, 7811. https://doi.org/10.1039/D3SC04585D
[9]. Poulson, B.G.; Alsulami, Q.A.; Sharfalddin, A.; El Agammy, E.F.; Mouffouk, F.; Emwas, A.-H.; Jaremko, L.; Jaremko, M. Cyclodextrins: Structural, Chemical, and Physical Properties, and Applications. Polysaccharides. 2022; 3, 1-31. https://doi.org/10.3390/polysaccharides3010001
[10]. Neaz, S., Alam, M.M., Imran, A.B. Advancements in cyclodextrin-based controlled drug delivery: Insights into pharmacokinetic and pharmacodynamic profiles. Heliyon, 2024; 10(e39917). https://doi.org/10.1016/j.heliyon.2024.e39917
[11]. Vinodh, M.; Alipour, F.H.; Mohamod, A.A.; Al-Azemi, T.F. Molecular Assemblies of Porphyrins and Macrocyclic Receptors: Recent Developments in Their Synthesis and Applications. Molecules, 2012; 17, 11763-11799. https://doi.org/10.3390/molecules171011763
[12]. Akkari, A. C. S., E. V. R. Campos, A. F. Keppler, L. F. Fraceto, E. D. Paula, G. R. Tófoli & D. R. D. Araujo. Budesonide-hydroxypropyl-β-cyclodextrin inclusion complex inbinary poloxamer 407/403 system for ulcerative colitis treatment: Aphysico-chemical study from micelles to hydrogels. Colloids and Surfaces B: Biointerfaces 138, 2016; 138–147. https://doi.org/10.1016/j.colsurfb.2015.11.048
[13]. Venuti, V., Cannavà, C., Cristiano, M. C., Fresta, M., Majolino, D., Paolino, D., Stancanelli, R., Tommasini, S & Ventura, C. A. A characterization study of resveratrol/sulfobutylether-β-cyclodextrin inclusion complex andin vitro anticancer activity. Colloids and Surfaces B: Biointerfaces, 2014; 115. 22–28. https://doi.org/10.1016/j.colsurfb.2013.11.025
[14]. Ha, W., Yu, J., Song, X. Y., Chen, J & Shi, Y. P. Tunable temperature-responsive supramolecular hydrogels formed by prodrugs as a codelivery system. ACS Applied Material Interfaces, 2014. 6(13). 10623–10630. https://pubs.acs.org/doi/10.1021/am5022864
[15]. Yakupova, L.R.; Skuredina, A.A.; Markov, P.O.; Le-Deygen, I.M.; Kudryashova, E.V. Cyclodextrin Polymers as a Promising Drug Carriers for Stabilization of Meropenem Solutions. Applied Science. 2023; 13, 3608. https://doi.org/10.3390/app13063608
[16]. Bai H, Wang J, Phan CU, Chen Q, Hu X, Shao G, Zhou J, Lai L, Tang G. Cyclodextrin-based host-guest complexes loaded with regorafenib for colorectal cancer treatment. Nature Communication, 2021;3;12(1):759. https://doi.org/10.1038/s41467-021-21071-0.
[17]. Khalid Q, Ahmad M, UsmanddMinhas M. Hydroxypropyl-β-cyclodextrin hybrid nanogels as nano-drug delivery carriers to enhance the solubility of dexibuprofen: Characterization, in vitro release, and acute oral toxicity studies. Advance Polymer Technology, 2018; 37: 21712185. https://doi.org/10.1002/adv.21876.
[18]. Rizvi S.S.B, Akhtar, N., Minhas, M.U., Mahmood A, Khan KU. Synthesis and Characterization of Carboxymethyl Chitosan Nanosponges with Cyclodextrin Blends for Drug Solubility Improvement. Gels, 2022 ;8(1):55. https://doi.org/10.3390/gels8010055
[19]. Li, H., Jie, Z., Caifen, W., Wei Q., Xiaoxiao, H., Jiabin, T., Lin, Y., Guoqing, Z., Xiaohong, R, Zegeng, L & Jiwen, Z. Paeonol loaded cyclodextrin metal-organic framework particles for treatment of acute lung injury via inhalation. International Journal of Pharmaceutics, 2020;587, 119649. https://doi.org/10.1016/j.ijpharm.2020.119649.
[20]. Wang, F., Bao, X., Fang, A., Li, H., Zhou, Y., Liu, Y., Jiang, C., Wu, J & Song, X. 2018. Nanoliposome-Encapsulated Brinzolamide-hydropropyl-β-cyclodextrin Inclusion Complex: A Potential Therapeutic Ocular Drug-Delivery System. Frontiers in Pharmacology, 2018; 9. https://doi.org/10.3389/fphar.2018.00091
[21]. Wang, C., Xiaojing, L., Shangyuan, S., David, J., McClements, Long, C., Jie, L., Aiquan, J., Jinpeng, W., Zhengyu, J & Chao, Q. Preparation, characterization and in vitro digestive behaviors of emulsions synergistically stabilized by γ-cyclodextrin/sodium caseinate/alginate. Food Research International, 2022;160. 111634. https://doi.org/10.1016/j.foodres.2022.111634
[22]. Welliver, M., & McDonough, J. P. Anesthetic Related Advances with Cyclodextrins. The Scientific World Journal, 2007;7,364–371. https://doi.org/10.1100/tsw.2007.83
[23]. Pedersen, C.J. Cyclic polyethers and their complexes with metal salts. Journal of the American Society of Chemistry, 1967; 89. 7017–7036. https://doi.org/10.1021/ja01002a035
[24]. Grobelny, Z., Stolarzewicz, A., Morejko-Buz, B., Bartsch, R. A., Yamato, K., Fernandez, F. A & Maercker, A. Preparation and Decomposition of Potassium Alkalide−Lipophilic Crown Ether Complexes in Tetrahydrofuran. Journal of Organic Chemistry, 2002;67(22). 7807–7812. https://dx.doi.org/10.1021/jo026086r
[25]. Qian, Y., Wu, Y., Qiu, S., He, X., Liu, Y., Kong, X. Y., Tian, W., Jiang, L & Wen, L. A. Bioinspired Free-Standing 2D Crown-Ether-Based Polyimine Membrane for Selective Proton Transport. Angewandte Chemie International Edition, 2023;16(8) 62. e202300167. https://doi.org/10.1002/anie.202300167
[26]. Lee, S. F., Zhu, X. M., Wang, Y. X. Xuan, S. H., You, Q., Chan, W. H., Wong, C. H., Wang, F., Yu, J. C & Cheng, C. H. Ultrasound, pH, and magnetically responsive crown-ether-coated core/shell nanoparticles as drug encapsulation and release systems. ACS Applied Materials & Interfaces Journal, 2013;5(5). 1566–1574. https://doi.org/10.1021/am4004705
[27]. You, X., Xiao-Jie, J., Fan, H., Yuan, W., Zhuang, L., Wei, W., Rui, X & Liang-Yin, C. Polymersomes with Rapid K+-Triggered Drug Release Behaviors. ACS Applied Materials & Interfaces, 2017; 9(22). https://doi.org/10.1021/acsami.7b05701
[28]. Angelini, G., Michela, P., Giovanna, M., Milvia, M & Carla, G. Neutral liposomes containing crown ether-lipids as potential DNA vectors. Biochimica et Biophysica Acta (BBA) – Biomembranes, 2013;1828(11). 2506-2512. https://doi.org/10.1016/j.bbamem.2013.06.003
[29]. Tasharrofi, N., Nourozi, M. & Ahmadifard, Z. Development and Optimization of a Novel Crown Ether-Incorporated Liposome for Improved Ocular Drug Delivery. BioNanoSciences, 2025; 15,261 https://doi.org/10.1007/s12668-025-01867-w
[30]. Chai, T., Mengtong, Z., Shuo, W., Jiankang, F., Xibin, F., Shihe, S., Chichong, L & Guofan, J. Curcumin/nido-carborane complexes incorporated with crown ether/sodium alginate encapsulated drug delivery strategies exhibit pH-responsive release and enhanced in vitro anti-tumor activity. Dyes and Pigments, 2024;231,112428. https://doi.org/10.1016/j.dyepig.2024.112428
[31]. Gao, Y.; Huang, Y.; Ren, C.; Xiong, S.; Guo, X.; Zhao, Z.; Guo, L.; Huang, Z. Construction of Cisplatin-18-Crown-6 Complexes Through Supramolecular Chemistry to Improve Solubility, Stability, and Antitumor Activity. International Journal of Molecular Sciences, 2024;25, 13411. https://doi.org/10.3390/ijms252413411.
[32]. Aragón, P. V., Mónica, R. J. F., Angélica, V. G & Miriam, E. F. Study of the stability of dopamine encapsulated in TiO2 and TiO2/15-crown-5 ether matrix. Ceramics International, 2024; 50(7). 10959-10966. https://doi.org/10.1016/j.ceramint.2023.12.412.
[33]. Goswami, A., Ai, K., Takashi, O., Hubiao, H & Takuzo, A. Fluoro-Crown Ether Phosphate as Efficient Cell-Permeable Drug Carrier by Disrupting Hydration Layer. Journal of the American Chemical Society, 2024;146 (33), 23406-23411. https://doi.org/10.1021/jacs.4c06464
[34]. Sultan, H., Arshad, N & Lateef, M. Novel Crown Ether-Functionalized Fusidic Acid Butyl Ester: Synthesis, Biological Evaluation, In Silico ADMET, and Molecular Docking Studies. Molecules, 2025;30(9), 2033. https://doi.org/10.3390/molecules30092033
[35]. Wang, X., Zheng, X., Liu, X., Zeng, B., Xu, Y., Yuan, C & Dai, L. K+-Responsive Crown Ether-Based Amphiphilic Copolymer: Synthesis and Application in the Release of Drugs and Au Nanoparticles. Polymers, 2022; 14 (3), 406. https://doi.org/10.3390/polym14030406
[36]. Bonnin, M.A., and Feldmann, C. Insights of the Structure and Luminescence of Mn2+/Sn2+-Containing Crown-Ether Coordination Compounds. Inorganic Chemistry, 2021;60(19), 14645–14654
https://doi.org/10.1021/acs.inorgchem.1c01662
[37]. Gokel, G. W., Barbour, L. J., Wall, S. L. D & Meadows, E. S. Macrocyclic polyethers as probes to assess and understand alkali metal cation-π-interactions. Coordination Chemistry Reviews, 2001; 222(1),127–154. http://dx.doi.org/10.1016/S0010-8545(01)00380-0
[38]. Gokel, G.W. Crown Ethers and Cryptands. Monograph in Supramolecular Chemistry; The Royal Society of Chemistry: London, England, 1991. https://doi.org/10.1039/9781788010917
[39]. Bell, M. M., Gutsche, N.T., King, A.P., Baidoo, K.E., Kelada, O.J., Choyke, P.L & Escorcia, F.E. Glypican-3-Targeted Alpha Particle Therapy for Hepatocellular Carcinoma. Molecules, 2020;26(4). https://doi.org/10.3390/molecules26010004
[40]. Chen, L., Zhang, H. Y. & Liu, Y. High affinity crown ether complexes in water: Thermodynamic analysis, evidence of crystallography and binding of NAD+. Journal of Organic Chemistry, 2012. 77. 9766–9773. https://doi.org/10.1021/jo301911w
[41]. You, X. R., Ju, X. J., He, F., Wang, Y., Liu, Z., Wang, W., Xie, R & Chu, L. Y. Polymersomes with Rapid K+-Triggered Drug Release Behaviors. ACS Applied Material Interfaces. 2017; 9, 22, 19258–19268. https://doi.org/10.1021/acsami.7b05701
[42]. Chehardoli. G., & A. Bahmani. The role of crown ethers in drug delivery. Supramolecular Chemistry, 2019; 31(4), 221-238, https://doi.org/10.1080/10610278.2019.1568432
[43]. Oral, I., Ott, F., Abetz, V. Thermodynamic study of crown ether–lithium/magnesium complexes based on benz-1,4-dioxane and its homologues. Physical Chemistry Chemical Physics, 2022; 24, 11687-11695, https://doi.org/10.1039/D2CP01076C
[44]. Monserrat, K., Gratzel, M & Tundo, P. Light-Induced Charge Injection in Functional Crown Ether Vesicles. Journal of the American Chemical Society, 1980; 102(17). 5521–5529. https://doi.org/10.1021/ja00537a018
[45]. Ullah, F.; Khan, T.A.; Iltaf, J.; Anwar, S.; Khan, M.F.A.; Khan, M.R.; Ullah, S.; Fayyaz ur Rehman, M.; Mustaqeem, M.; Kotwica-Mojzych, K.; et al. Heterocyclic Crown Ethers with Potential Biological and Pharmacological Properties: From Synthesis to Applications. Applied Sciences, 2022; 12, 1102. https://doi.org/10.3390/app12031102
[46]. Echegoyen, L. E., Hernandez, J.C., Kaifer, A.E., Gokel, G. W & Echegoyen, L. Aggregation of Steroidal Lariat Ethers: The First Example of Nonionic Liposomes (Niosomes) Formed from Neutral Crown Ether Compounds. Journal of the Chemical Society, Chemical Communication, 1988; 836–837. https://doi.org/10.1039/c39880000836
[47]. Muheyati, M., Wu, G., Li, Y., Pan, Z., & Chen, Y. Supramolecular nanotherapeutics based on cucurbiturils. Journal of nanobiotechnology, 2024; 22(1), 790. https://doi.org/10.1186/s12951-024-03024-z
[48]. Behrend, R., E. Meyer & F. Rusche. Ueber Condensationsproducte RUS Glycoluril und formaldehyd. Justus Liebigs Annalen der Chemie, 1905; 339, 1. http://dx.doi.org/10.1002/jlac.19053390102
[49]. Freeman, W. A., Mock, W. L & Shih, N. Y. Cucurbituril. Journal of the Americal Chemical Society, 1981;103. 24. 7367–7368. https://doi.org/10.1021/ja00414a070
[50]. Ma, W. J., Chen, J. M., Jiang, L., Yao, J & Lu, T. B. The delivery of triamterene by cucurbit [7]uril: Synthesis, structures and pharmacokinetics study. Molecular Pharmaceutics, 2013; 10, 4698–4705. https://doi.org/10.1021/mp400529m
[51]. Wu, D., Li, Y., Yang, J., Shen, J., Zhou, J., Hu, Q., Yu, G., Tang, G & Chen, X. Supramolecular Nanomedicine Constructed from Curcurbit[8]uril-Based Amphiphilic Brush Copolymer for Cancer Therapy. ACS Applied Material Interfaces. (2017). 9. 51. 44392–44401. https://doi.org/10.1021/acsami.7b16734
[52]. Klarić, D.; Borko, V.; Parlov Vuković, J.; Pilepić, V.; Budimir, A.; Galić, N. Host–Guest Interactions of Cucurbit[7]uril with Nabumetone and Naproxen: Spectroscopic, Calorimetric, and DFT Studies in Aqueous Solution. Molecules. 2025, 30, 2558. https://doi.org/10.3390/molecules30122558
[53]. Kim, E., D. Kim, H. Jung, J. Lee, S. Paul, N. Selvapalam, Y. Yang, N. Lim, C. G. Park & K. Kim. 2010. Facile, Template-Free Synthesis of Stimuli-Responsive Polymer Nanocapsules for Targeted Drug Delivery. Angewandte Chemie International Edition. 14: 49(26): 4405-8. https://doi.org/10.1002/anie.201000818
[54]. Webber, M. J., E.A. Appel, B. Vinciguerra, A.B. Cortinas, L.S. Thapa, S. Jhunjhunwala, L. Isaacs, R. Langer, & D.G. Anderson. 2016. Supramolecular PEGylation of biopharmaceuticals, Proceedings of the National Academy of Sciences of the United States of America. 113 (50) 14189-14194, https://doi.org/10.1073/pnas.1616639113.
[55]. Chen, H., Yueyue, C., Han, W., Jiang-Fei, X., Zhiwei, S & Xi, Z. Supramolecular polymeric chemotherapy based on cucurbit[7]uril-PEG copolymer. Biomaterials, 2018; 178 697-705. https://doi.org/10.1016/j.biomaterials.2018.02.051.
[56]. Yue, L., C. Sun, T. H. C. H. T. Kwong & R. Wang. 2020. Cucurbit[7]uril-functionalized magnetic nanoparticles for imaging-guided cancer therapy. Journal of Material Chemistry B, 2020, 8, 2749. https://doi.org/10.1039/D0TB00306A
[57]. Cheng, Q., M. Xu, C. Sun, K. Yang, Z. Yang, J. Li, J. Zheng, Y. Zheng & R. Wang. 2022. Enhanced antibacterial function of a supramolecular artificial receptor-modified macrophage (SAR-Macrophage). Material Horizon, 2022, 9, 934. https://doi.org/10.1039/D1MH01813B
[58]. Ding, Y., Jianwen, W., Xingping, Q., Wenting, G., Long, X., Ying, Z., Yonghua, Z., Jingwei, L., Shengke, L., Greta, S. P. M & Ruibing, W. Hyaluronic acid-based supramolecular medicine with polyamines sequestration capability for cooperative anti-psoriasis. Carbohydrate Polymers, 2022; 296. 119968. https://doi.org/10.1016/j.carbpol.2022.119968.
[59]. Vinciguerra, B., P. Y. Zavalij & L. Isaacs. 2015. Synthesis and Recognition Properties of Cucurbit[8]uril Derivatives. ACS Organic Letters, 2015; 17(20). https://doi.org/10.1021/acs.orglett.5b02558
[60]. Das, D., Assaf, K.I., Nau, W.M. Applications of Cucurbiturils in Medicinal Chemistry and Chemical Biology. Frontiers in Chemistry. 2019;7. https://doi.org/10.3389/fchem.2019.00619
[61]. Kim, K., Selvapalam, N & Oh, D. H. Cucurbiturils—A New Family of Host Molecules. Journal of Inclusion Phenomena and macrocyclic chemistry, 2004; 50, 31–36. http://dx.doi.org/10.1007/s10847-004-8835-7
[62]. Liu, Y. H., Zhang, Y. M., Yu, H. J & Liu, Y. Cucurbituril-Based Biomacromolecular Assemblies. Angewandte Chemie International Edition, 2020; 60(8), 3870-3880 https://doi.org/10.1002/anie.202009797
[63]. Zhang, Y. Q., Zeng, J. P., Zhu, Q. J., Xue, S. F & Tao, Z. Molecular capsules formed by three different cucurbit [5]urils and some lanthanide ions. Journal of Molecular Structure, 2009; 929(1-2) 167–173. https://doi.org/10.1016/j.molstruc.2009.04.022
[64]. Zhang, X., Xu, X., Li, S., Wang, L. H., Zhang, J & Wang, R. A systematic evaluation of the biocompatibility of cucurbit[7]uril in mice. Scientific Reports, 2018; 8. 8819. https://doi.org/10.1038/s41598-018-27206-6
[65]. Lazar, A. I., Biedermann, F., Mustafina K. R., Assaf, K. I., Hennig, A & Nau, W. M. Nanomolar Binding of Steroids to Cucurbit[n]urils: Selectivity and Applications. Journal of the American Chemical Society, 2016; 138 (39), 13022–13029. https://doi.org/10.1021/jacs.6b07655
[66]. Plumb, J. A., B. Venugopal, R. Oun, N. Gomez-Roman, Y. Kawazoe, N. S. Venkataramanan & N. J. Wheate. Cucurbit[7]uril encapsulated cisplatin overcomes cisplatin resistance via a pharmacokinetic effect. Metallomics, 2012; 4 (6), 561. https://doi.org/10.1039/c2mt20054f
[67]. Zhang, X., Xu, X., Li, S., Li, L., Zhang, J & Wang, R. A Synthetic Receptor as a Specific Antidote for Paraquat Poisoning. Theranostics, 2019; 9. 633–645. https://doi.org/10.7150/thno.31485
[68]. Kuok, K. I., In Ng, P. C., Ji. X., Wang. C., Yew. W. W., Chan. D. P. C., Zheng, J., Lee, S. M. Y & Wang, R. Supramolecular strateg for reducing the cardiotoxicity of bedaquiline without compromising its antimycobacterial efficacy. Food and Chemical Toxicology, 2018; 119. 425–429. https://doi.org/10.1016/j.fct.2017.12.022
[69]. Basilotta, R., Mannino, D., Filippone, A., Casili, G., Prestifilippo, A., Colarossi, L., Raciti, G., Esposito, E., Campolo, M. Role of Calixarene in Chemotherapy Delivery Strategies. Molecules, 2021; 26, 3963. https://doi.org/10.3390/molecules26133963
[70]. Nimse, S. B., & T. Kim. Biological applications of functionalized calixarenes. Chemical Society Reviews, 2013; 42(1). 366-386. https://doi.org/10.1039/C2CS35233H
[71]. Guo, D. S., & Y. Liu. Supramolecular Chemistry of p-Sulfonatocalix[n]arenes and Its Biological Applications. Accounts of Chemical Research, 2014; 47(7) 1925. https://doi.org/10.1021/ar500009g
[72]. Ukhatskaya, E. V., S. V. Kurkov, S. E. Matthews & T. Loftsson. Encapsulation of Drug Molecules into Calix[n]arene Nanobaskets. Role of Aminocalix[n]arenes in Biopharmaceutical Field. Journal of Pharmaceutical Sciences, 2013; 102(10). 3485. https://doi.org/10.1002/jps.23681
[73]. Rodik, R. V., V. I. Boyko & V. I. Kalchenko. Calixarenes in Bio-Medical Researches. Current Medicinal Chemistry, 2009; 16(13), 1630-1655. https://doi.org/10.2147/DDDT.S83213
[74]. Mokhtari, B & K. Pourabdollah. Applications of calixarene nano-baskets in pharmacology. Journal of Inclusion Phenomena and Macrocyclic Chemistry, 2012; 73(1). https://doi.org/10.1007/s10847 011 0062 z
[75]. Bagnacani, V., V. Franceschi, M. Bassi, M. Lomazzi, G. Donofrio, F. Sansone, A. Casnati & R. Ungaro. Arginine clustering on calix[4]arene macrocycles for improved cell penetration and DNA delivery. Nature Communications, 2013; 4(1721). https://doi.org/10.1038/ncomms2721
[76]. Curtis, A. D. M, & Hoskins, C. Simple Calix[n]arenes and Calix [4]resorcinarenes as Drug Solubilizing Agents Journal of Nanomedicine Research, 2015; 2(28). https://doi.org/10.15406/jnmr.2015.02.00028
[77]. Li, B., Meng, Z., Li, Q., Huang, X., Kang, Z., Dong, H., Chen, J., Sun, J., Dong, Y., Li, J., Jia, X., Sessler, J. L., Meng, Q & Li, C. A pH responsive complexation-based drug delivery system for oxaliplatin. Chemical Science, 2017; 8. 4458–4464. https://doi.org/10.1039/C7SC01438D
[78]. Geraci, C., Consoli, G. M. L., Giusappe, G., Galante, E., Palmigiano, A., Pappalardo, M. S., Puma, S. D & Spadaro, A. The first self-adjuvant multicomponent potential vaccine candidates by tethering of four or eight MUC1 antigenic immunodominant PDTRP units on a calixarene platform: synthesis and biological evaluation. Bioconjugate Chem, 2013; 24. 10. 1710–1720. https://doi.org/10.1021/bc400242y
[79]. Ostos, F. J., J. A. Lebron, M. L. Moya, M. Lopez, A. Sanchez, A. Clavero, C. B. Garcia-Calderon, I. V. Rosado & P. L. Cornejo. P-Sulfocalix[6]arene as Nanocarrier for Controlled Delivery of Doxorubicin. Chemistry – An Asian Journal, 2017; 12. 6. 679-689. https://doi.org/10.1002/asia.201601713.
[80]. Zhao, Z.M., Wang. Y., Han, J., Zhu, H.D., An, L. Preparation and Characterization of Amphiphilic Calixarene Nanoparticles as Delivery Carriers for Paclitaxel. Chemical & Pharmaceutical Bulletin. 2015;63(3):180-186. https://doi.org/10.1248/cpb.c14-00699
[81]. Mo, J., Eggers, P.K., Yuan, Z., Raston. C.L., Lim, L.Y. Paclitaxel-loaded phosphonated calixarene nanovesicles as a modular drug delivery platform. Scientific Reports, 2016; 6, 23489. https://doi.org/10.1038/srep23489.
[82]. Yerga, L. G., I. Posadas, C. D. L. Torre, J. R. Almansa, F. Sansone, C. O. Mellet, A. Casnati, J. M. G. Fernandez & V. Cena. Docetaxel-Loaded Nanoparticles Assembled from β-Cyclodextrin/Calixarene Giant Surfactants: Physicochemical Properties and Cytotoxic Effect in Prostate Cancer and Glioblastoma Cells. Frontiers in Pharmacology, 2017; 8: 249. https://doi.org/10.3389/fphar.2017.00249
[83]. Mo, J., P. K. Eggers, X. Chen, M. R. H. Ahamed, T. Becker, L. Y. Lim & C. L. Raston. Shear induced carboplatin binding within the cavity of a phospholipid mimic for increased anticancer efficacy. Natural Scentific. Reports, 2015; 5:10414. https://doi.org/10.1038/srep10414
[84]. Renziehausen, A., A. D. Tsiailanis, R. P. Evgenios, K. Stylos, C. Chatzigiannis, K. O'Neill, T. Crook, A. G. Tzakos, N. Syed. 2019. Encapsulation of Temozolomide in a Calixarene Nanocapsule Improves Its Stability and Enhances Its Therapeutic Efficacy against Glioblastoma. Molecular Cancer Therapeutics, 2019; 18 (9): 1497–1505. https://doi.org/10.1158/1535-7163.MCT-18-1250
[85]. Wang, Y. X., D. S. Guo, Y. C. Duan, Y. J. Wang & Y. Liu. 2015. Amphiphilic p-Sulfonatocalix[4]arene as “Drug Chaperone” for Escorting Anticancer Drugs. Scientific Reports, 2015; 5, 9019. https://doi.org/10.1038/srep09019.
[86]. Sreedevi, P., Jyothi, B. N., Manu, M. J., Vishnu, P. M., Cherumuttathu, H. S., Varma R. L., Kaustabh, K. M. Dynamic self-assembly of mannosylated-calix[4]arene into micelles for the delivery of hydrophobic drugs. Journal of Controlled Release, 2021; 339. 284-296. https://doi.org/10.1016/j.jconrel.2021.09.038
[87]. Drakalska, E., Denitsa, M., Yana, M., Dessislava, B., Georgi, M., Margarita, G., Liudmil, A., Nikolay, L & Stanislav, R. Hybrid liposomal PEGylated calix[4]arene systems as drug delivery platforms for curcumin. International Journal of Pharmaceutics. 2014; 472(1-2), 165-174, https://doi.org/10.1016/j.ijpharm.2014.06.034
[88]. Bandela, A. K., V. K. Hinge, D. S. Yarramala & C. P. Rao. Versatile, Reversible, and Reusable Gel of a Monocholesteryl Conjugated Calix[4]arene as Functional Material to Store and Release Dyes and Drugs Including Doxorubicin, Curcumin, and Tocopherol. ACS Applied Materials & Interfaces, 2015; 7(21), 25946024. https://doi.org/10.1021/acsami.5b02506
[89]. Zhang, T. X., Zhang, Z. Z., Yue, Y. X., Hu, X. Y., Huang, F., Shi, L., Liu, Y & Guo, D. S. A General Hypoxia-Responsive Molecular Container for Tumor-Targeted Therapy. Advanced Materials, 2020; 32(28), 1908435. https://doi.org/10.1002/adma.201908435
[90]. Zyryanov, G. V., Kopchuk, D. S., Kovalev, I. S., Santra, S., Majee, A & Ranu, B. C. Pillararenes as Promising Carriers for Drug Delivery. International Journal of Molecular Sciences, 2023; 24 (6), 5167. https://doi.org/10.3390/ijms24065167
[91]. Ogoshi, T., S. Kanai, S. Fujinami, T. A. Yamagishi & Y. Nakamoto. para-Bridged Symmetrical Pillar[5]arenes: Their Lewis Acid Catalyzed Synthesis and Host–Guest Property. Journal of the American Chemical Society, 2008; 130 (15) 5022. https://doi.org/10.1021/ja711260m
[92]. Xue, M., Yang, Y., Chi, X., Zhang, Z & Huang, F. Pillararenes, A New Class of Macrocycles for Supramolecular Chemistry. Accounts of Chemical Research, 2012 ; 45(8). 1294–1308. https://doi.org/10.1021/ar2003418
[93]. Lu, B., Xia, J., Huang, Y & Yao, Y. The design strategy for pillararene based active targeted drug delivery systems. Chemical Communications, 2023; 59 (81), 12091–12099. https://doi.org/10.1039/D3CC04021F
[94]. Shangguan, L., Chen, Q., Shi, B & Huang, F. Enhancing the solubility and bioactivity of anticancer drug tamoxifen by water-soluble. Chemical Communications, 2017; 53(70), 9749-9752. https://doi.org/10.1039/C7CC05305C
[95]. Wheate, N. J., Dickson, K. A., Kim, R.R., Nematollahi, A., Macquart, R,B., Kayser, V., Yu, G., Church, W.B., & Marsh D.J. Host-Guest Complexes of Carboxylated Pillar[n]arenes With Drugs. Journal of Pharmaceutical Sciences, 2016. 105. 3615. https://doi.org/10.1016/j.xphs.2016.09.008
[96]. Sun, Y. L., Yang, Y. W., Chen, D. X., Wang, G., Zhou, Y., Wang, C. Y & Stoddart, J. F. Mechanized silica nanoparticles based on pillar [5]arenes for on-command cargo release. Small, 2013;9.3224–3229.x. https://doi.org/10.1002/smll.201300445
[97]. Barbera, L., D. Franco, L. M. D. Plano, G. Gattuso, S. P. P. Guglielmo, G. Lentini, N. Manganaro, N. Marino, S. Pappalardo, M. F. Parisi, F. Puntoriero, I. Pisagatti & A. Notti. 2017. Awater-soluble pillar[5]arene as a new carrier for an old drug. Organic and Biomolecular Chemistry, 2017;15, 3192-3195. https://doi.org/10.1039/C7OB00530J
[98]. Jain, A., Prajapati, S.K., Kumari, A., Mody, N., Bajpai, M. Engineered nanosponges as versatile biodegradable carriers: An insight. Journal of Drug Delivery Science and Technology, 2020; 57. 101643. https://doi.org/10.1016/j.jddst.2020.101643
[99]. Yu, G., Dan, W., Yang, L., Zhihua, Z., Li, S., Jiong, Z., Qinglian, H., Guping, T & Feihei, H. 2016. A pillar[5]arene-based [2]rotaxane lights up mitochondria. Chemical Science, 2016, 7, 3017-3024. https://doi.org/10.1039/C6SC00036C
[100]. Yang, K., Yincheng, C., Jia, W., Yuchao, L., Yuxin, P., Shoupeng, C., Feng, W & Zhichao, P. Supramolecular Vesicles Based on Complex of Trp-Modified Pillar[5]arene and Galactose Derivative for Synergistic and Targeted Drug Delivery. Chemistry of Materials, 2016; 28(7).1990-1993. https://doi.org/10.1021/acs.chemmater.6b00696
[101]. Wang, Y., Ming, J., Zelong, C., Xianjun, H., Liang, P., Zhichao, P & Yuxin, P. 2020. Tumor microenvironment responsive supramolecular glyco-nanovesicles based on diselenium-bridged pillar[5]arene dimer for targeted chemotherapy. Chemical Communications, 2020, 56(73), 10642.https://doi.org/10.1039/D0CC04149A
[102]. Silva, A. F. M. D., Nathalia, M. D. C., Tamires, S. F., Isabela, A. A. B., Dayenny, L. D., Carlos, A. S., Matheus, L. C., Vanessa, N., Antonio, P. J., Braulio, S. A., Luis, F. R. P., Thiago, C. D & Celia, M. R. 2022. Responsive Supramolecular Devices Assembled from Pillar[5]arene Nanogate and Mesoporous Silica for Cargo Release. Applied Nano Materials ACS, Mater, 2022; 5 (10), 13805–13819.https://doi.org/10.1021/acsanm.2c01408
[103]. Ding, C., Ying, L., Ting, W & Jiajun, F. Triple-stimuli-responsive nanocontainers assembled by water-soluble pillar[5]arene-based pseudorotaxanes for controlled release. Journal of Materials Chemistry B, 2016; 4(16), 2819-2827. https://doi.org/10.1039/C6TB00459H
[104]. Lan S,, Liu, Y., Shi, K & Ma, D. Acetal-Functionalized Pillar[5]arene: A pH-Responsive and Versatile Nanomaterial for the Delivery of Chemotherapeutic Agents. ACS Appl Bio Mater. 2020;3(4):2325-2333. https://doi.org/10.1021/acsabm.0c00086
[105]. Huang, X., Zhang, X., Qian, T., Ma, J., Cui, L & Li, C. Synthesis of a water-soluble 2,2'-biphen[4]arene and its efficient complexation and sensitive fluorescence enhancement towards palmatine and berberine. Beilstein Journal of Organic Chemistry, 2018; 14: 2236-2241. https://doi.org/10.3762/bjoc.14.198
Published
Versions
- 2026-01-20 (2)
- 2025-06-30 (1)