Effectiveness of Kepok Banana (Musa paradisiaca) Bread-Based Adsorbent for Fe(III) Removal Using HCl Activation: Freundlich and Langmuir Isotherm Models
DOI:
https://doi.org/10.22437/chp.v9i1.44926Keywords:
Activated carbon, kepok banana stem, iron (Fe)Abstract
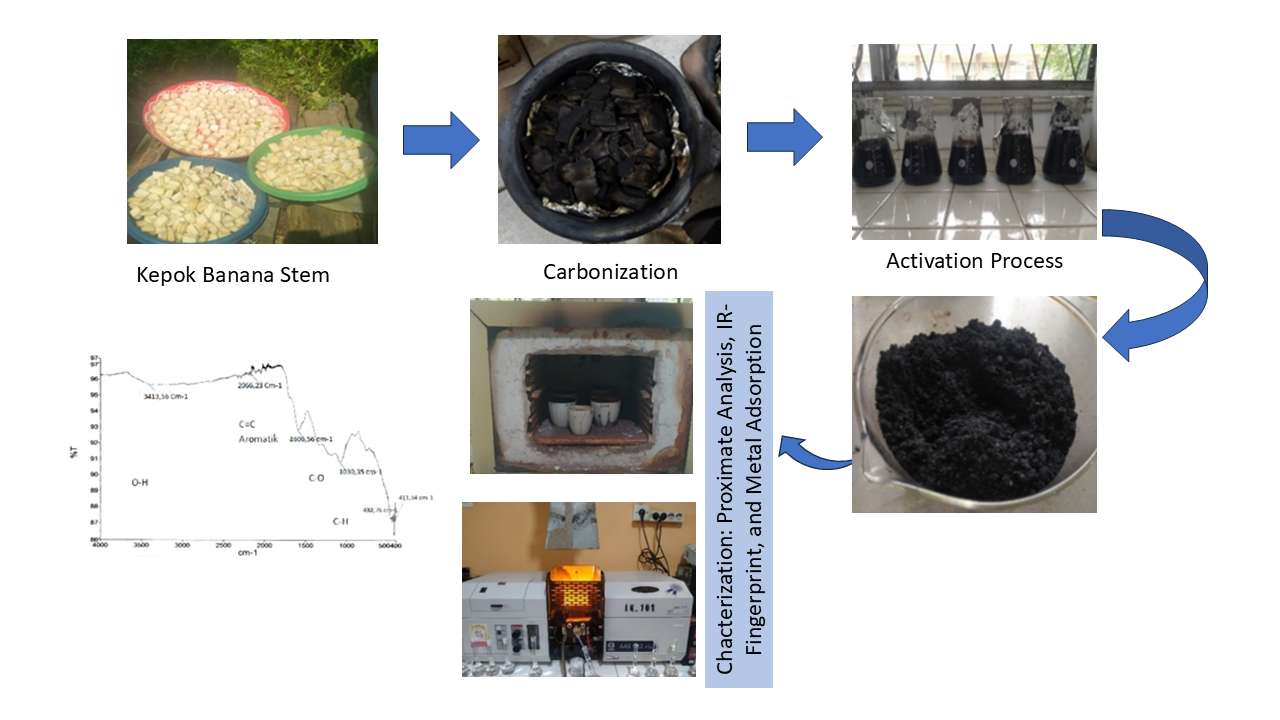
Activated carbon can be synthesized from lignocellulosic biomass, such as kepok banana (Musa paradisiaca) stems, which are an abundant agricultural waste rich in cellulose (~64%). In this study, kepok banana stems were utilized as a precursor to produce activated carbon using hydrochloric acid (HCl) as the chemical activating agent. The activation process employed HCl concentrations of 1.5, 2.0, 3.0, 5.0, and 7.0 N, with a carbonization temperature of 400 °C for 1 hour and an Fe adsorption contact time of 30 minutes. The resulting activated carbon was evaluated based on its physicochemical properties according to the Indonesian National Standard (SNI 06-3730-1995). The sample treated with 3.0 N HCl showed optimal characteristics, including moisture content of 2.34%, ash content of 0.28%, volatile matter content of 2.05%, and fixed carbon content of 95.33%. Its iodine number reached 1116.98 mg/g, and the Fe ion removal efficiency was 99.14%. FTIR spectroscopy confirmed the presence of functional groups typical of activated carbon—O–H, aromatic C=C, C–H, and C–O—suggesting good adsorption potential. Furthermore, adsorption behavior was analyzed using the Freundlich isotherm model, which describes multilayer adsorption on heterogeneous surfaces. The findings demonstrate that activated carbon derived from kepok banana stems is an effective, low-cost, and environmentally friendly adsorbent for iron removal, suitable for applications in water purification and wastewater treatment.
Downloads
References
[1] Sujiono EH, Zabrian D, Zurnansyah M, Mulyati M, Zharva V, Samnur S, Humairah. Fabrication and Characterization of Coconut Shell Activated Carbon Using Variation Chemical Activation for Wastewater Treatment Application. Result in Chemistry. 2022;4, 100291. https://doi.org/10.1016/j.rechem.2022.100291
[2] Haryati S, Yulhan AT, Asparia L. Preparation of Activated Carbon from Gelam Bark (Melaleuca Leucadendron) Which Originated from Tanjung Api-Api South Sumatra. Chemical Engineering Journal. 2017;23(2):77-86. https://doi.org/10.36706/jtk.v23i2.1564
[3] Ashari AW & Sitorus S. Effect of Time Variation on Xylene Using Activated Carbon from Banana Stem Waste (Musa Paradisiaca L). Atomic Journal. 2021;5(2):62-66.
[4] Deepa B, Abraham E, Cherian BM, Bismarck A, Blaker JJ, Pothan LA, Kottaisamy M. Structure, Morphology and Thermal Characteristics of Banana Nano Fibers Obtained by Steam Explosion. Bioresource Technology. 2011;102(2):1988-1997.
[5] Campbell NA, Reece JB. 2008. Biology. 8th Ed. Pearson Benjamin Cummings.
[6] Udyani K, Purwaningsih DY, Setiawan R, Yahya K. Preparation of Activated Carbon from Mangrove Charcoal using Combined Chemical and Physical Activation with Microwave. Journal of Science and Technology. 2019;23(1):39-46.
[7] National Standardization Agency. SNI 06-3730-1995:Test Method for Moisture Content in Solid Materials [indonesian national standard]. BSN 1995.
[8] Foo KY & Hameed BH. Insights into The Modeling of Adsorption Isotherm Systems. Chemical Engineering Journal. 2010;156(1):2-10. https://doi.org/10.1016/j.cej.2009.09.013
[9] Metcalf & Eddy. 2014. Wastewater Engineering: Treatment and Resource Recovery. 5th Ed. Mcgraw-hill education.
[10] Atkins P & Depaula J. 2010. Physical Chemistry. 9th ed. Oxford University Press.
[11] Do DD. 1998. Adsorption Analysis: Equilibria and Kinetics. Imperial College Press London
[12] Setianingsih T. 2020. Characterization Methods for Carbon Nanomaterials [Metode Karakterisasi Karbon Nanomaterial]. Universitas Brawijaya Press.
[13] National Standardization Agency. SNI 06-3730-1995: Test Method for Ash Content of Solid Materials [Indonesian National Standard]. BSN 1995.
[14] National Standardization Agency. SNI 06-3730-1995: Test method for volatile matter content in solid materials [Indonesian National Standard]. BSN 1995.
[15] National Standardization Agency. SNI 06-3730-1995: Test method for bound carbon content in solid materials [Indonesian National Standard]. BSN 1995.
[16] Kartika SE & Amran MB. Synthesis and Characterization of Poly (Anthranilic Acid-Co-Formaldehyde) for Adsorption of Pb(II) ion. Alchemy. 2021;9(1):15-25.
[17] Syahrir I, Samosir D, Destarini NA, Bariah B. Utilization of Banana Stem Waste (Musa Paradisiaca L) as Activated Charcoal Through Activation Process Using NaOH Activator. National Seminar on Research & Community Service Results (SNP2M) 2020; 54-59.
[18] Aryani F. Application of Physical Activation and Chemical Activation Methods in the Preparation of Activated Charcoal from Coconut Shells (Cocos Nucifera L). Indonesian Journal of Laboratory. 2019;1(2):16-20.
[19] Elfia M. Biosorption of Pb(II) Heavy Metal Ions using Kepok Banana Stem Biosorbent (Musa Acuminata Balbisiana Colla). Clinical Science: Journal of Health Analysts. 2019;7(2):76-82.
[20] Jankowska H, Swiatkowski A, Choma J. 1991. Active Carbon. T.J. Kemp, Editor. Ellis Horwood Ltd New York.
[21] Putri RW, Haryati S, Ramatullah R. Effect of Carbonation Temperature on the Quality of Activated Carbon from Sugarcane Bagasse Waste. Journal of Chemical Engineering University of Sriwijaya. 2019; 25(1):1-4.
[22] Rowe RC, Sheskey, PJ, Owen SC. 2006. Carboxy Methyl Cellulose Sodium. Handbook of Pharmaceutical Excipients. Fifth Edition. American Pharmacist Association Washington, 120–122.
[23] Amehaa B, Nadew TT, Tedla TS, Getye B, Mengie DA, Ayalneh S.
The Use of Banana Peel as a Low-Cost Adsorption Material for Removing Hexavalent Chromium from Tannery Wastewater: Optimization, Kinetic and Isotherm Study, and Regeneration Aspects. Royal Society of Chemistry. 2024;14(6):3617-4300. https://doi.org/10.1039/d3ra07476e
[24] Chafidz A, Astuti W, Augustia VAS, and Rofiah N. Removal of Methyl Violet Dye Via Adsorption using Activated Carbon Prepared from Randu Sawdust (Ceiba Pentandra). J. IOP Conference Series:Earth and Environmental Science. 2018;167(1):012013. https://doi.org/10.1088/1755-1315/167/1/012013
[25] Rahmawati MA, Salam AN, Munte WN, Chitraningrum N, and Rahmasari L. Synthesis and Characterization Analysis of Banana Peel Carbon Activated as Adsorption of Copper (Cu). J. Jurnal Ilmu Lingkungan. 2025;23(3):647-657.
[26] Cao Y, Zhang J, Yang X, and Shen D. Adsorption Behavior of Functional Groups in Activated Carbon Derived from Biomass. J. Journal of Environmental Chemical Engineering. 2020;8(3):103673. https://doi:10.1016/j.jece.2020.103673
Published
Versions
- 2026-01-20 (2)
- 2025-06-30 (1)